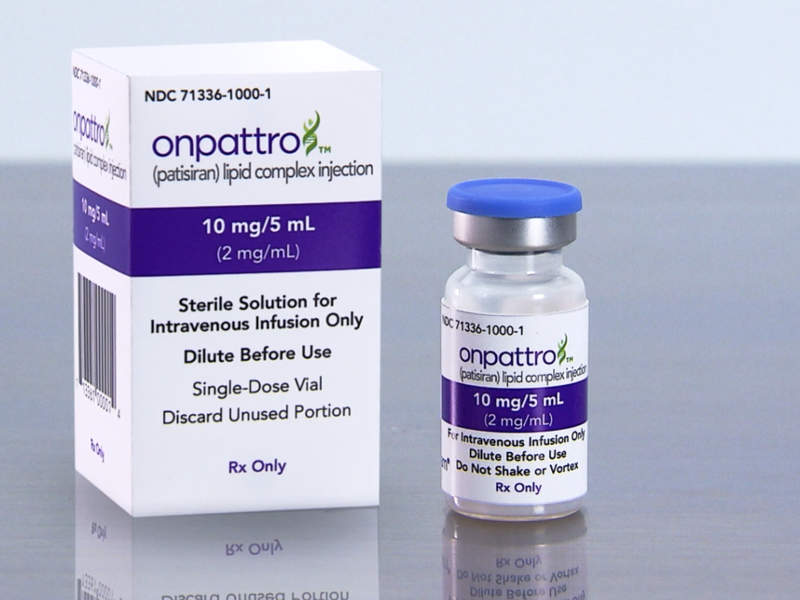
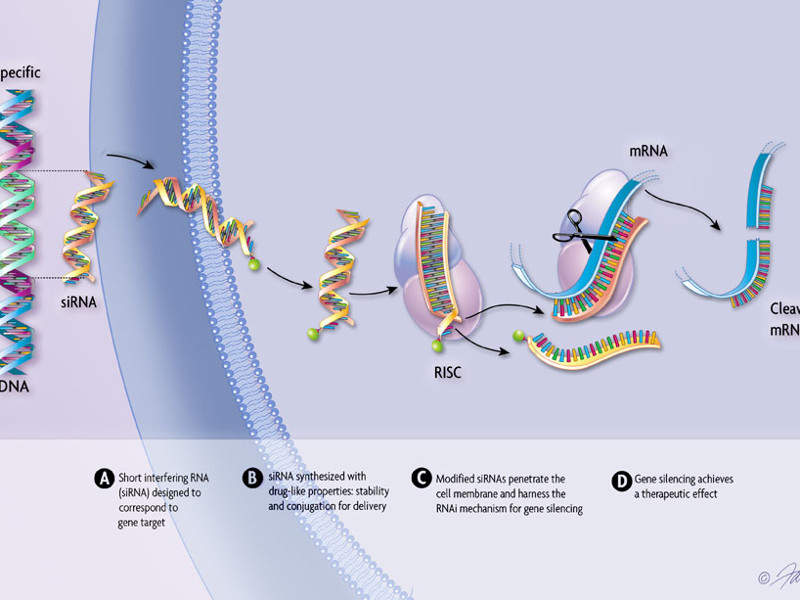
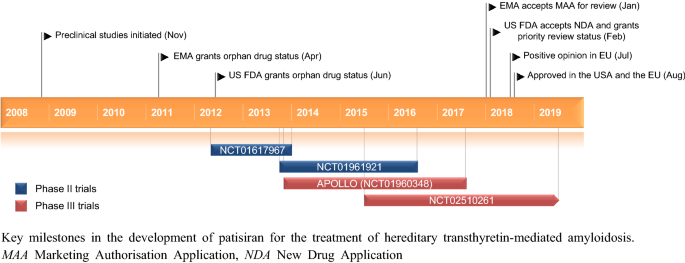
Patisiran, an RNAi therapeutic for the treatment of hereditary transthyretin-mediated amyloidosis | Neurodegenerative Disease Management

Brain Sciences | Free Full-Text | Advances in Treatment of ATTRv Amyloidosis: State of the Art and Future Prospects
Indirect treatment comparison of the efficacy of patisiran and inotersen for hereditary transthyretin-mediated amyloidosis with
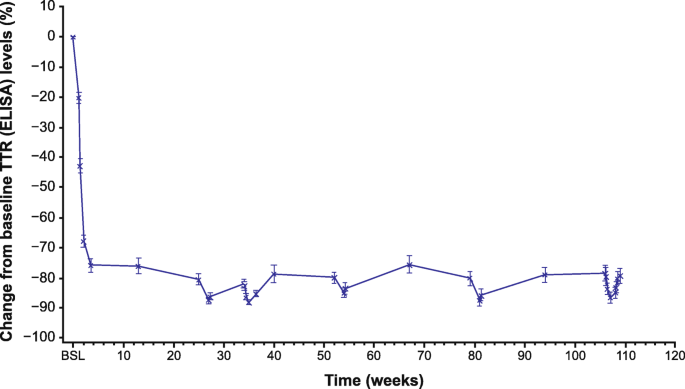
A phase II, open-label, extension study of long-term patisiran treatment in patients with hereditary transthyretin-mediated (hATTR) amyloidosis | Orphanet Journal of Rare Diseases | Full Text

PDF) Pharmacokinetics of Patisiran, the First Approved RNA Interference Therapy in Patients With Hereditary Transthyretin‐Mediated Amyloidosis

Alnylam Announces First-Ever FDA Approval of an RNAi Therapeutic, ONPATTRO™ (patisiran) for the Treatment of the Polyneuropathy of Hereditary Transthyretin-Mediated Amyloidosis in Adults | Business Wire

PDF) Patisiran, an RNAi therapeutic for the treatment of hereditary transthyretin-mediated amyloidosis