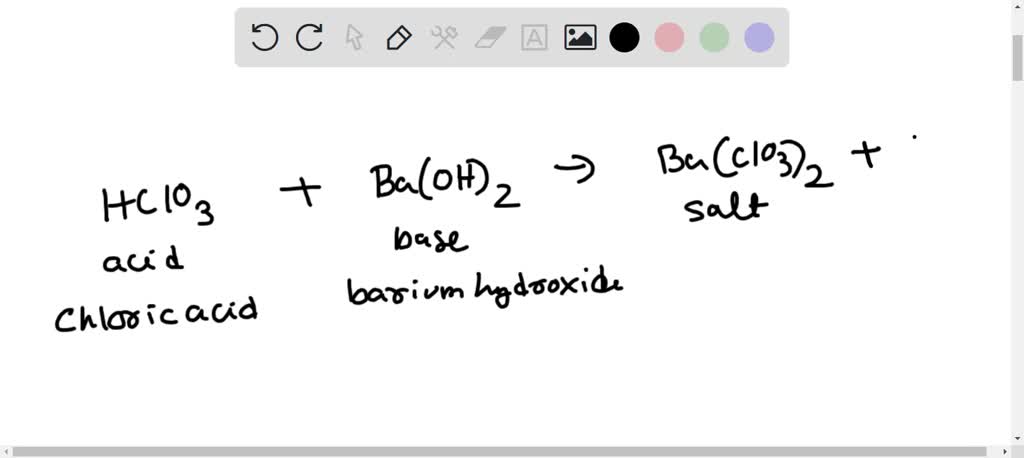
SOLVED: How many mL of 1.271 M HClO3 are required to completely neutralize 0.5151 g of Ca(OH)2 (MM = 74.09 )?
SOLVED: 0.570 g of HClO3 in 2.00 L of solution.Express the pH of the solution to three decimal places.

For the reaction Ba(OH)(2)+2HClO(3)rarr Ba(ClO(3))(2)+2H(2)O. Calculate the number of moles of H(2)O formed when 0.1 mole of Na(OH)(2) is treated with 0.0250 mole HClO(3).