EU Medicines Agency on Twitter: "How does EMA support accelerated development and approval of safe, effective & high-quality💊 and #vaccines against #COVID19? #RollingReview is one of the fast-track procedures EMA uses so

EU Medicines Agency on Twitter: "‼️ EMA has started a rolling review of #COVID19Vaccine HIPRA, intended for use as a #boosterdose in people who have already been fully vaccinated with a different #

ATAGI statement and clinical guidance on AstraZeneca COVID-19 vaccine following European Medicines Agency (EMA) safety review | Australian Government Department of Health and Aged Care

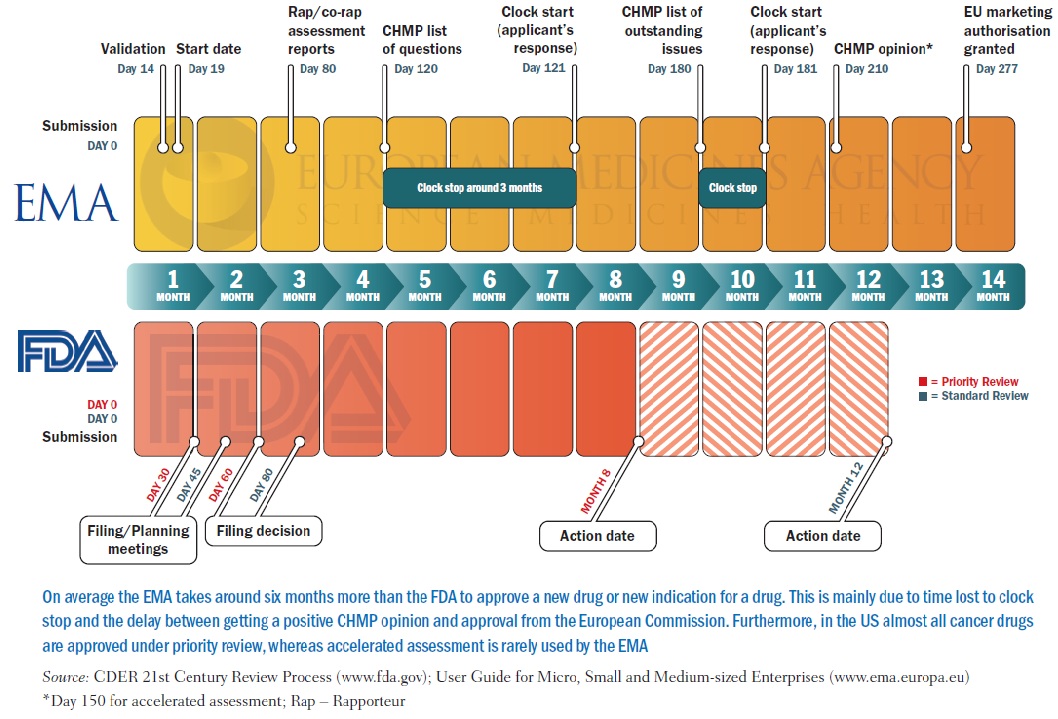
EMA public stakeholder meeting COVID 19 - EU's regulatory process for evaluation and approval - YouTube