
Network Analysis for Signal Detection in Spontaneous Adverse Event Reporting Database: Application of Network Weighting Normalization to Characterize Cardiovascular Drug Safety | springermedizin.de
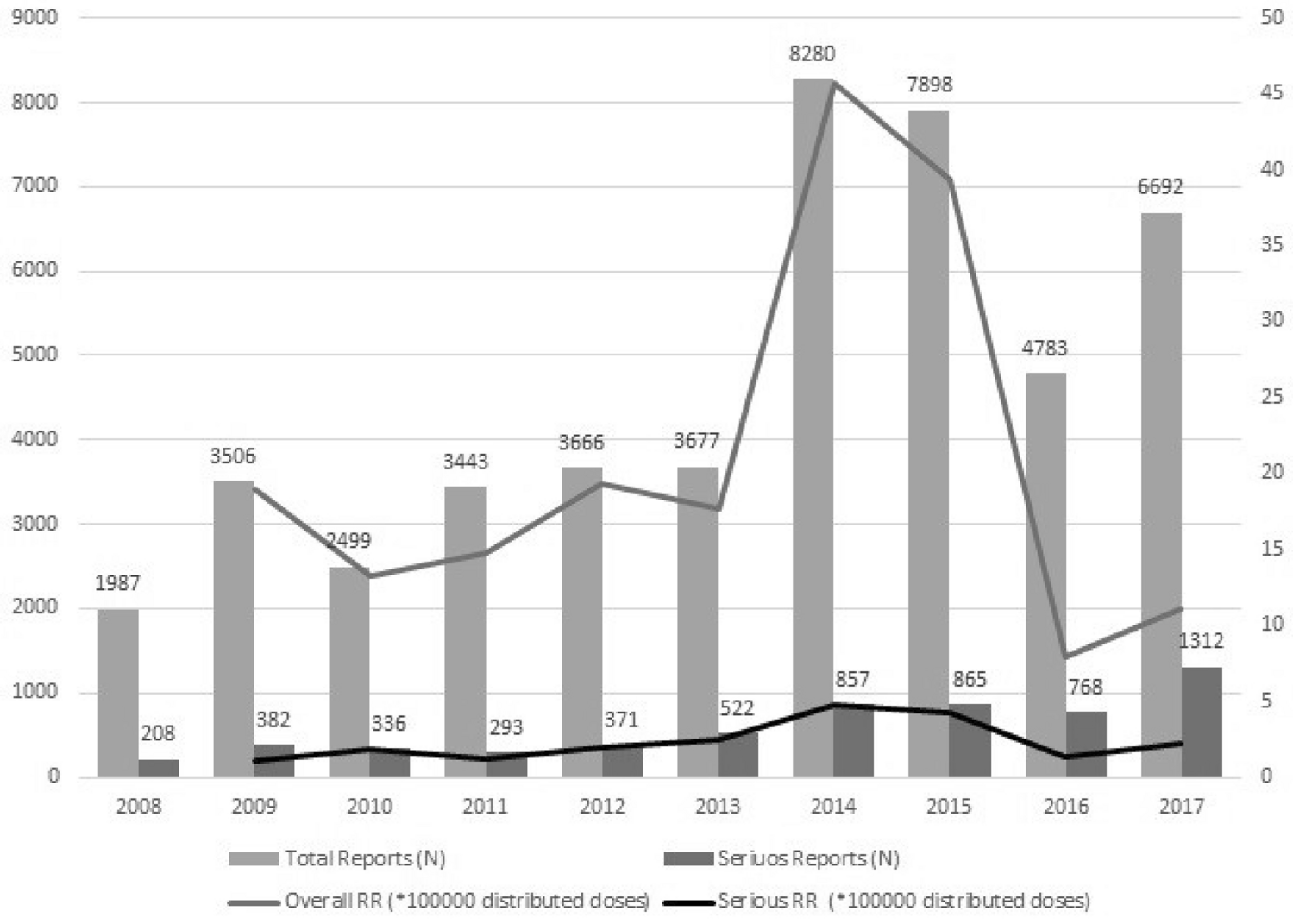
Ten years of vaccinovigilance in Italy: an overview of the pharmacovigilance data from 2008 to 2017 | Scientific Reports
Background of IME list • IME List survey results • Considerations in maintaining MedDRA- based term lists
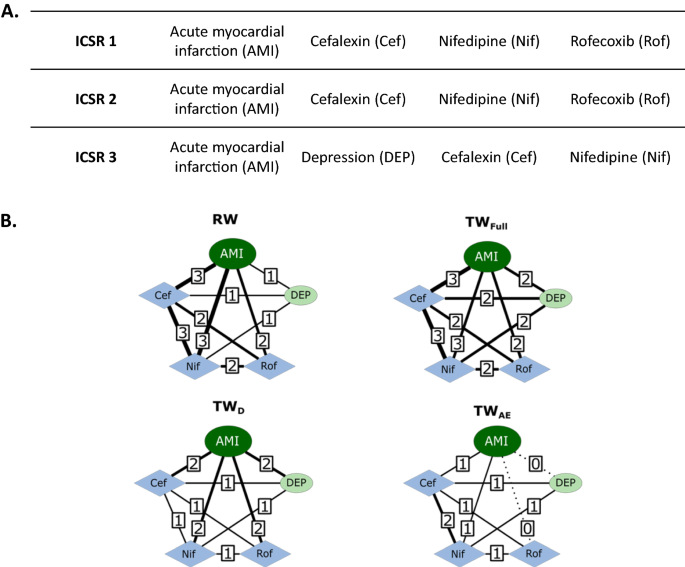
Network Analysis for Signal Detection in Spontaneous Adverse Event Reporting Database: Application of Network Weighting Normalization to Characterize Cardiovascular Drug Safety | SpringerLink
Background of IME list • IME List survey results • Considerations in maintaining MedDRA- based term lists

6. Perceived Gap Analysis and Research Needs | Current Hazardous Materials Transportation Research and Future Needs |The National Academies Press

PDF) Health horizons: Future trends and technologies from the European Medicines Agency's horizon scanning collaborations

Definitions and difference in PVG terms: Designated medical event (DME), Important medical events (IMEs), WHO critical terms and AESIs

Access-2-Healthcare SG - EMA Releases EudraVigilance List of Important Medical Event Terms This is a list of important medical event (IME) terms for users of its EudraVigilance safety database, intended to support
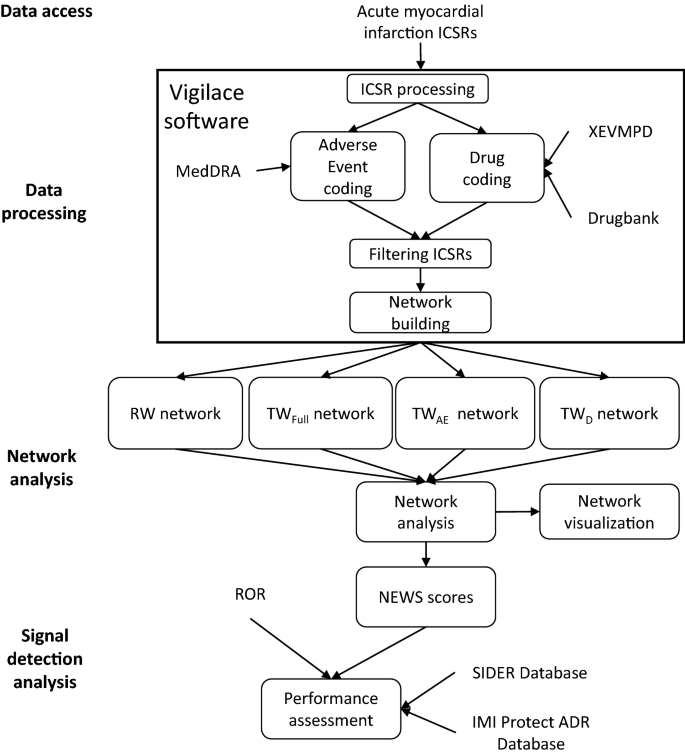
Network Analysis for Signal Detection in Spontaneous Adverse Event Reporting Database: Application of Network Weighting Normalization to Characterize Cardiovascular Drug Safety | SpringerLink







.jpg)



![PDF] Screening for adverse reactions in EudraVigilance | Semantic Scholar PDF] Screening for adverse reactions in EudraVigilance | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/747c892608ff2264a343509c19f362b2bad013f0/34-Table6-1.png)