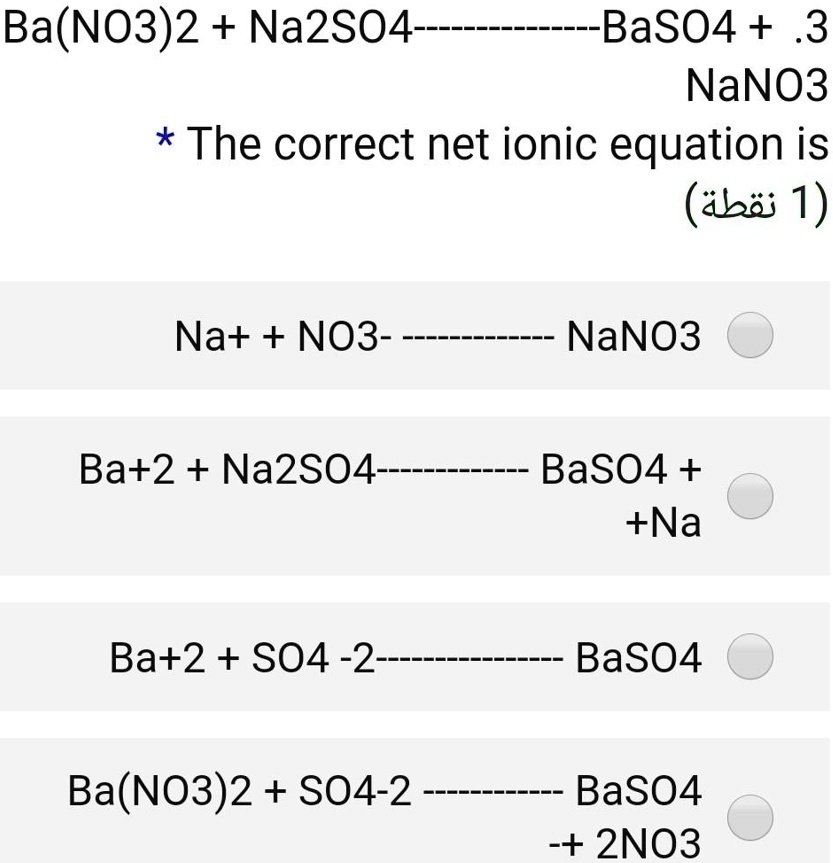
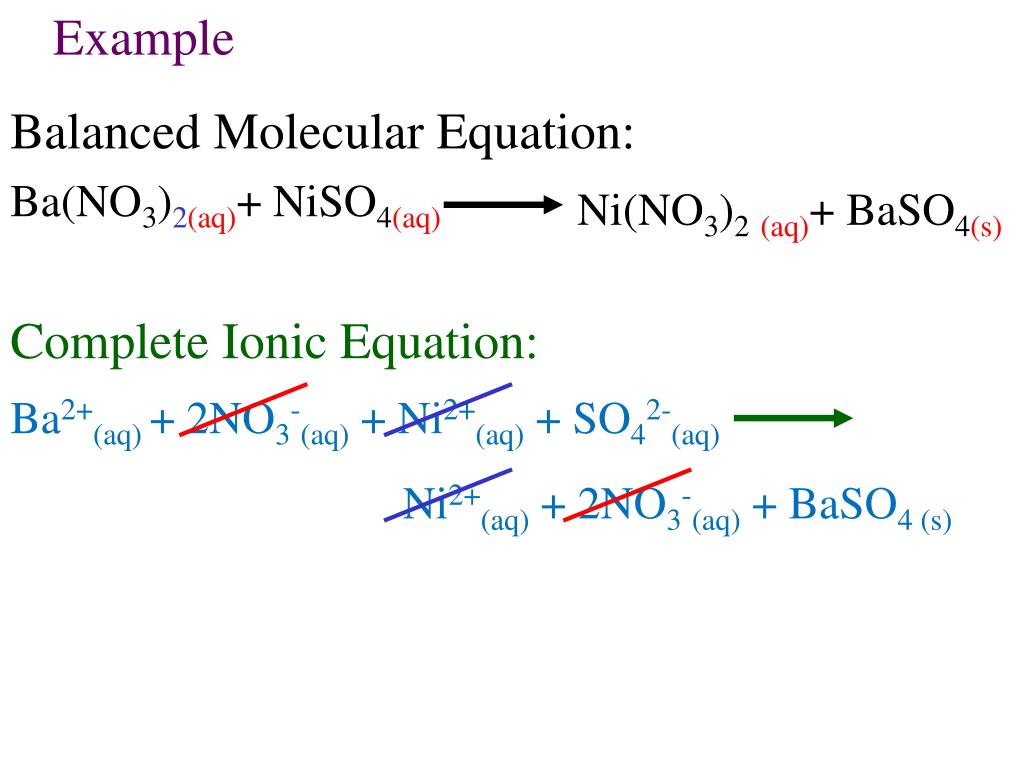
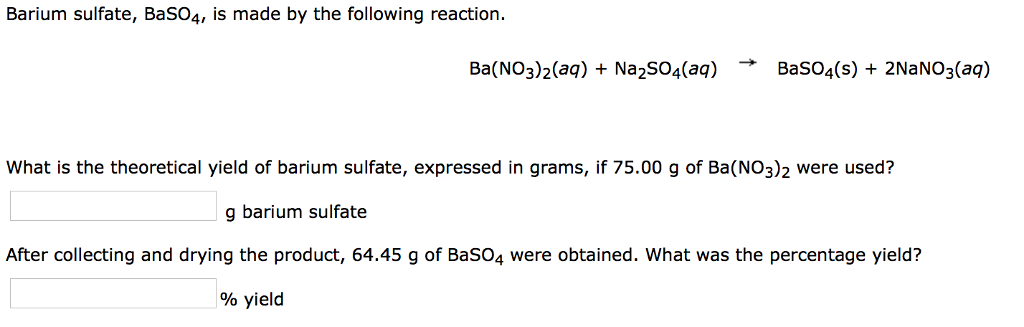Выполните полные и краткие ионные уравнения : 1) Na2SO4 + Ba (NO3)2 => 2) CuSO4 +Zn - Школьные Знания.com
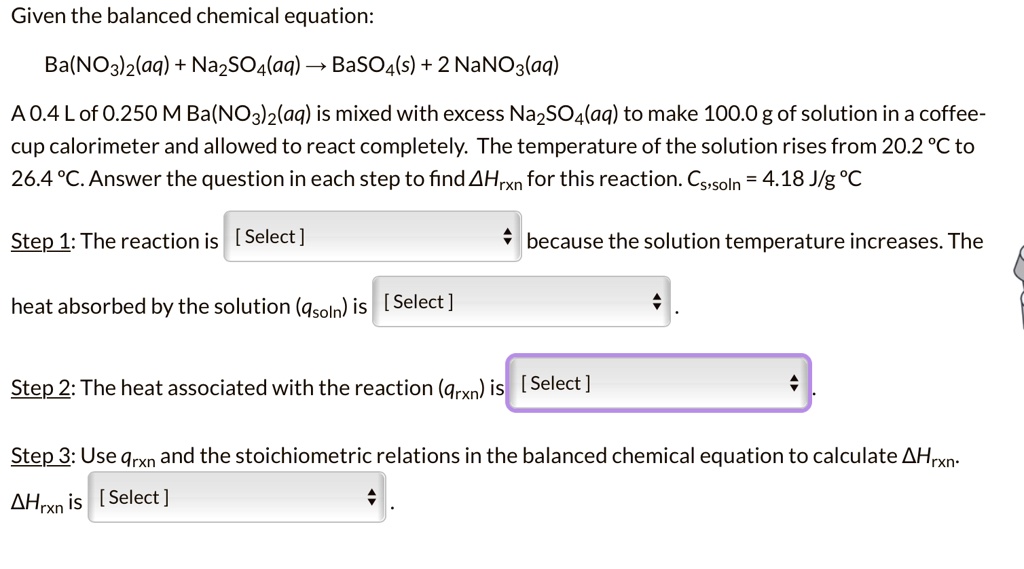
SOLVED: Given the balanced chemical equation: Ba(NO3)2(aq) + NazSO4(aq) 4 BaSO4(s) + 2 NaNOa(aq) A0.4Lof 0.250 M Ba(NO3)z(aq) is mixed with excess NazSOa(aq) to make 100.0 gof solution in a coffee- cup
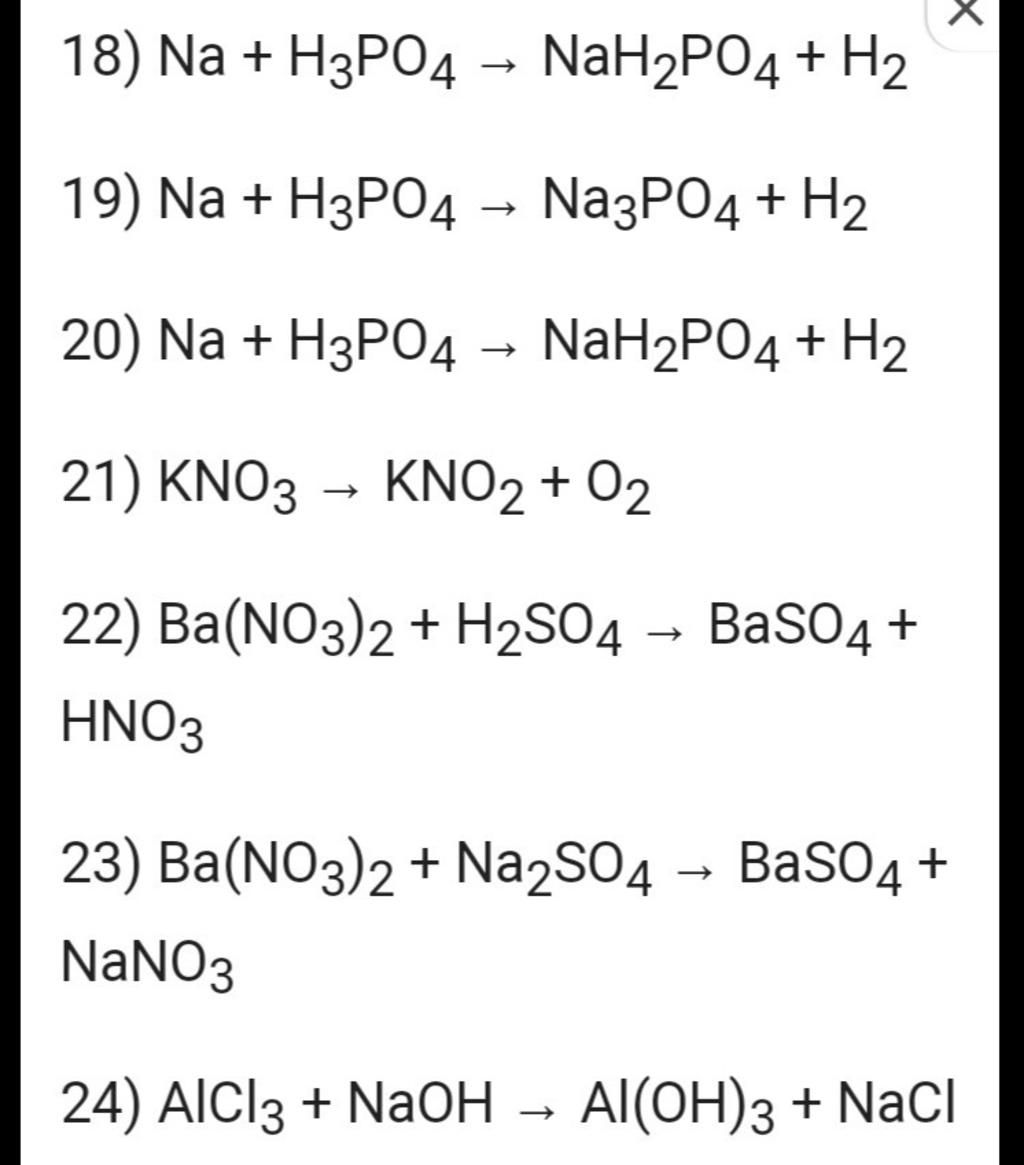
18) Na + H3PO4 → NaH₂PO4 + H₂ 19) Na + H3PO4 Na3PO4 + H2 20) Na + H3PO4 → NaH₂PO4 + H₂ H2 21) KNO3 → KNO2 + 02 22) Ba(NO3)2 + H₂SO4 → BaSO4 + HNO3 23) Ba(N

SOLVED: When 1.00 L of 1.00 M Ba(NO3)2 solution at 25.0 oC is mixed with 1.00 L of 1.00 M Na2SO4 solution at 25.0 oC in a coffee-cup calorimeter, the white solid

Ba(NO3)2+Na2So4= CuSO4+NaOH= Cu(OH)2+HNO3= Осуществите такие химические превращения: - Школьные Знания.com

Chem.docx - 1. Given The Balanced Chemical Equation: Ba No3 2 Aq Na2so4 Aq → Baso4 S 2 Nano3 Aq A 0.30 L Of 0.15 M Ba No3 2 Aq Is Mixed With - CHEMLOL666 | Course Hero